E2EDigitalPharma.info = End-to-End (E2E) Pharma Digitalization Resource Hub

System/Data Visuals
eCTD 3.2.2 vs. 4.0, Reference: https://www.ijdra.com/index.php/journal/article/view/788
"Use a picture. It's worth a thousand words." 1911, Arthur Brisbane
Elucidating Diagrams/Visuals for Programs/Initiatives, Systems/Data, with reference links provided below:
eCTD 3.2.2 vs. 4.0 (A table not a visual per se!)
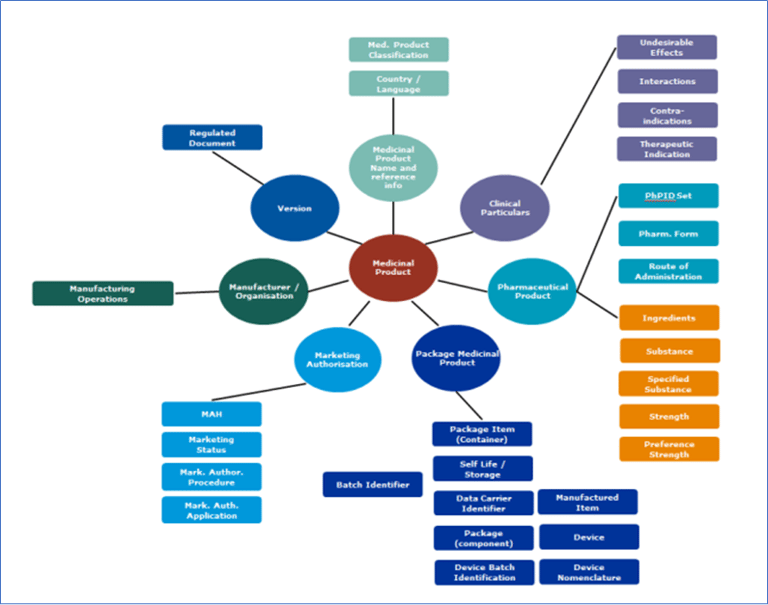
ICH IDMP
ICH IDMP, Overview of ISO data elements
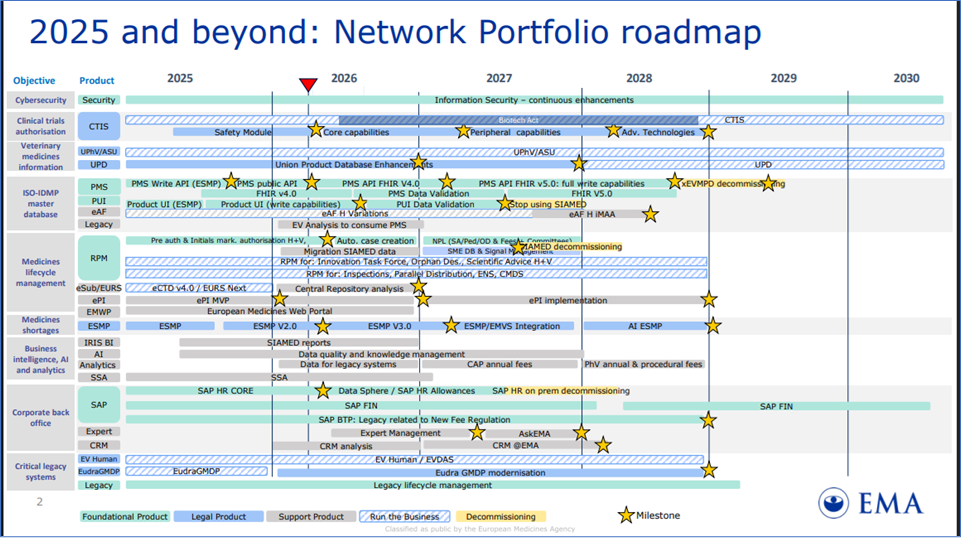
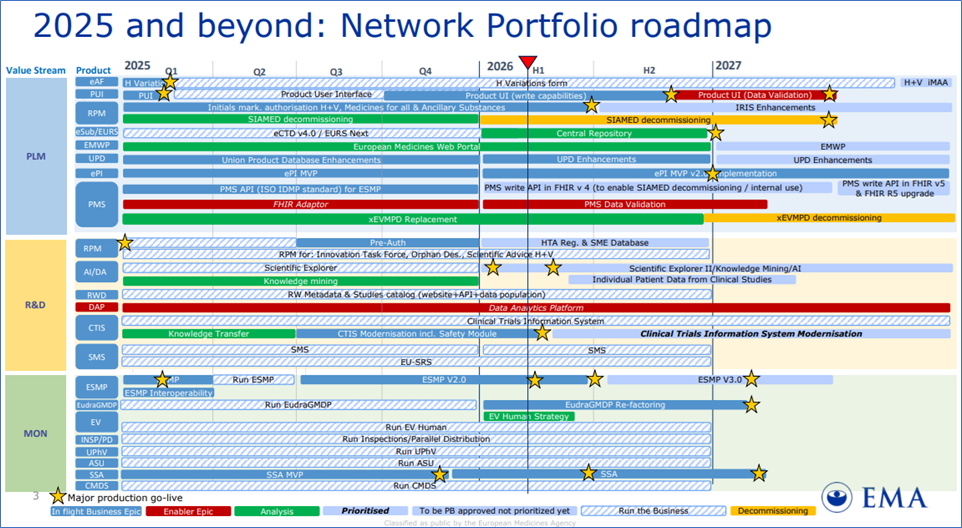
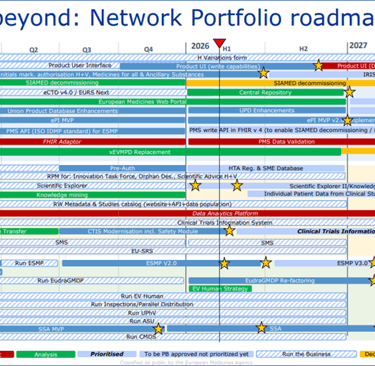
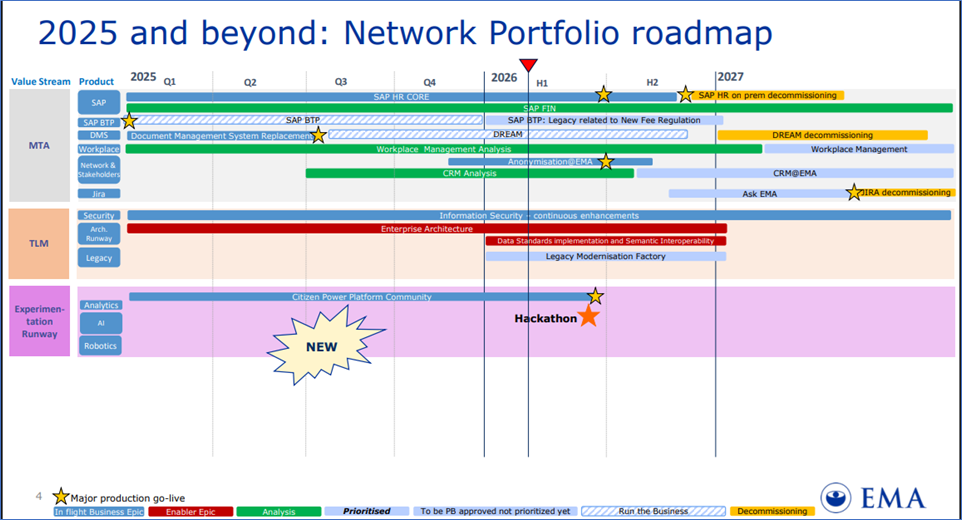
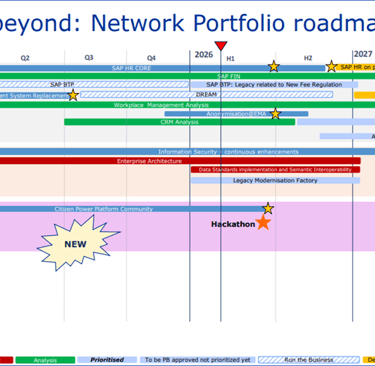
EMA Network Portfolio roadmap
PQ/CMC FHIR resources
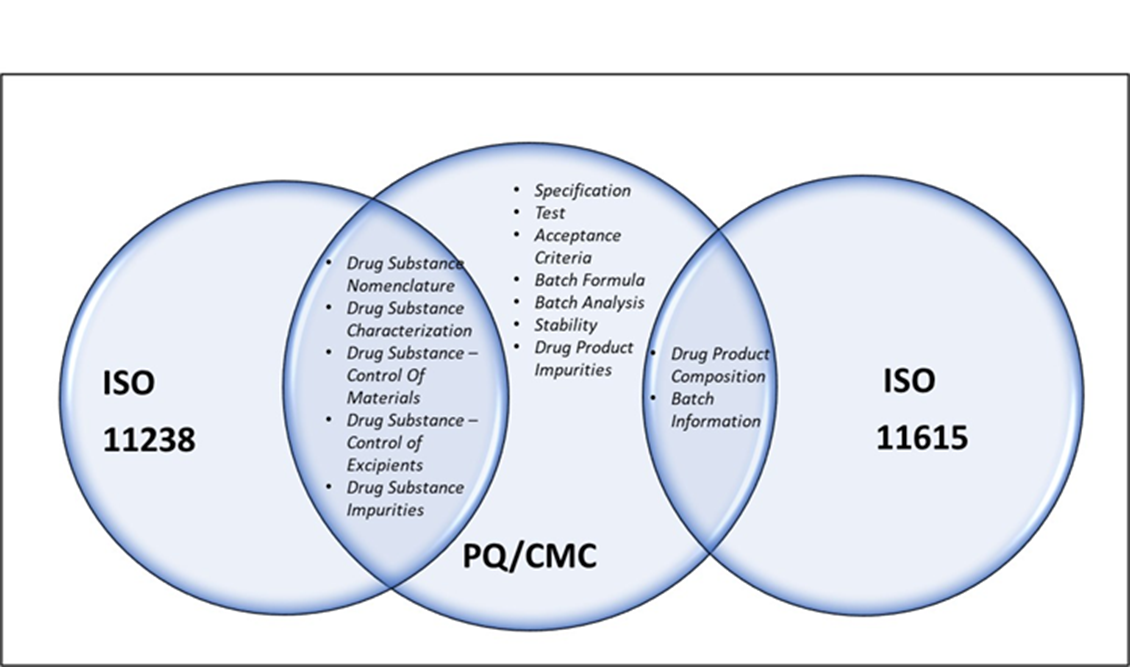
PQ/CMC and IDMP High-level mapping
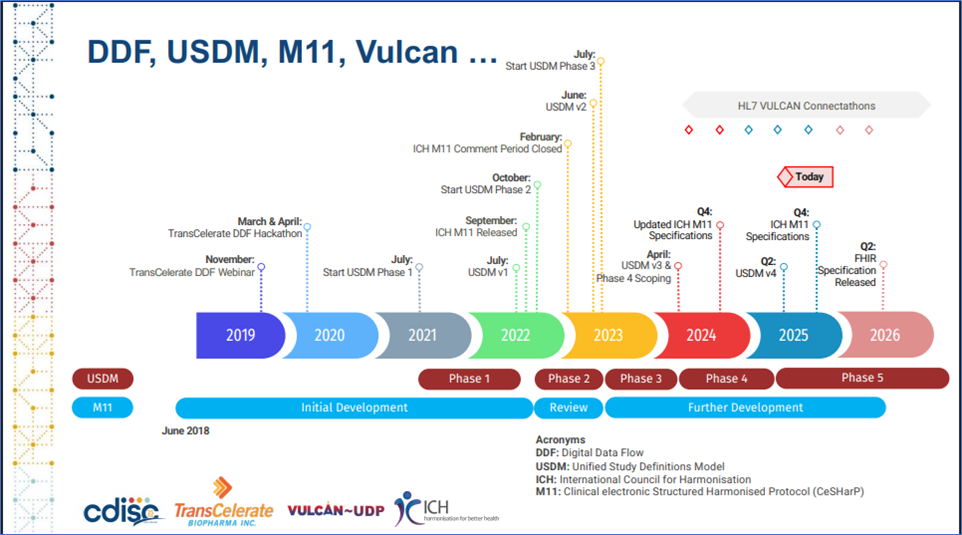
Digital Data Flow (DDF), Unified Study Definitions Model (USDM), ICH M11 Timeline
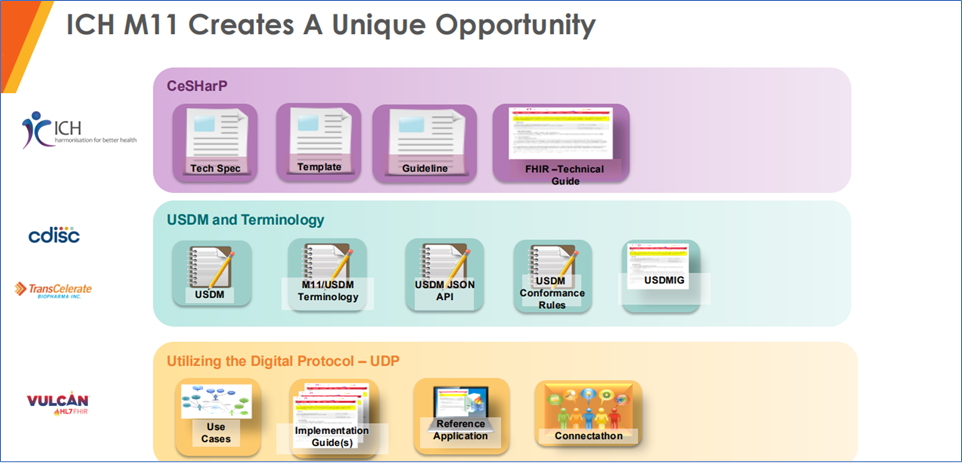
ICH M11 opportunity
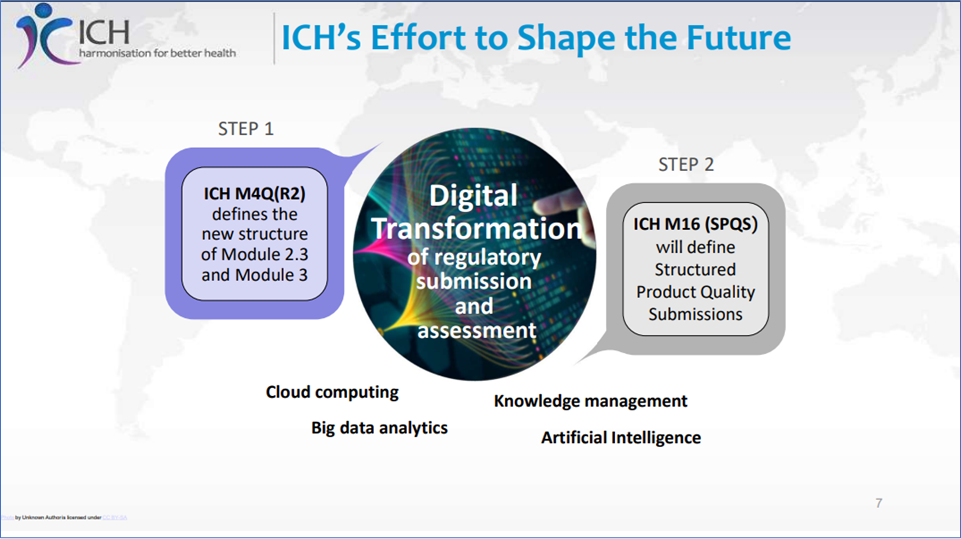
ICH M4Q(R2) to Structured Product Quality Submission (SPQS)(M16)
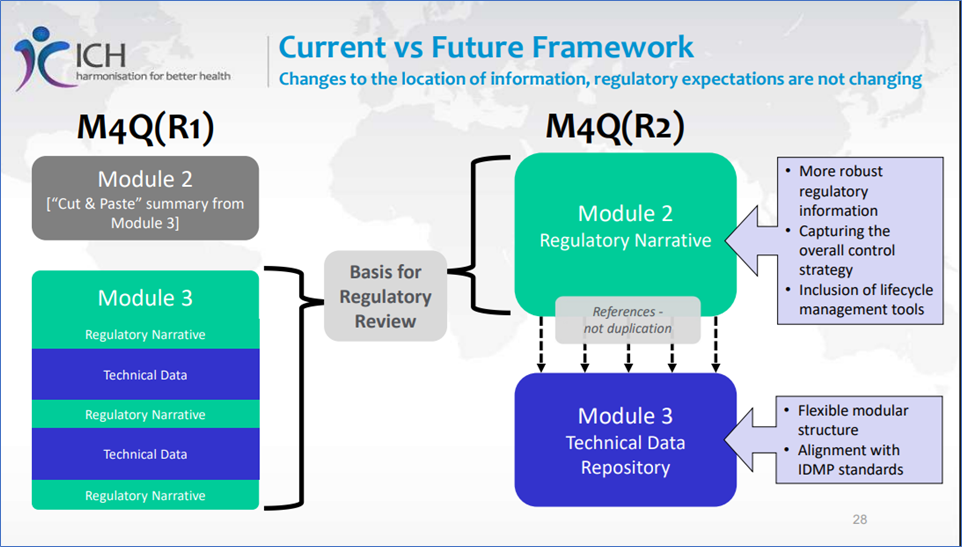
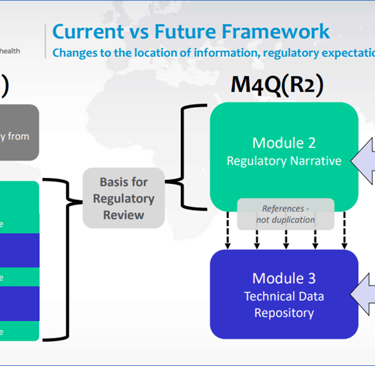
ICH M4Q(R1) vs. ICH M4Q(R2)
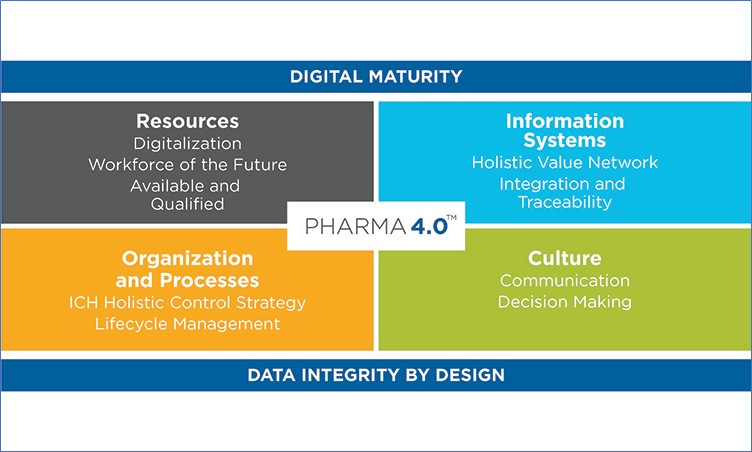
ISPE Pharma 4.0 ™ Operating Model (Another table, not a visual per se!)
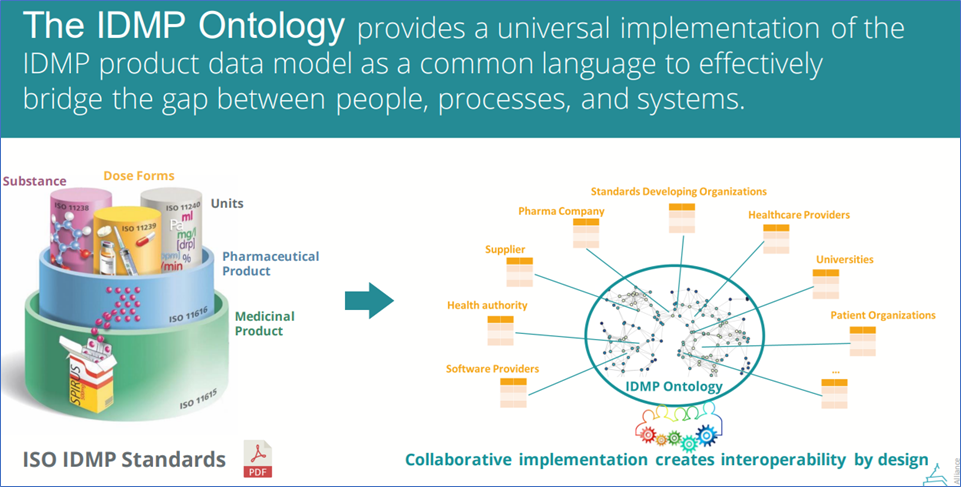
Pistoia Alliance IDMP-ontology
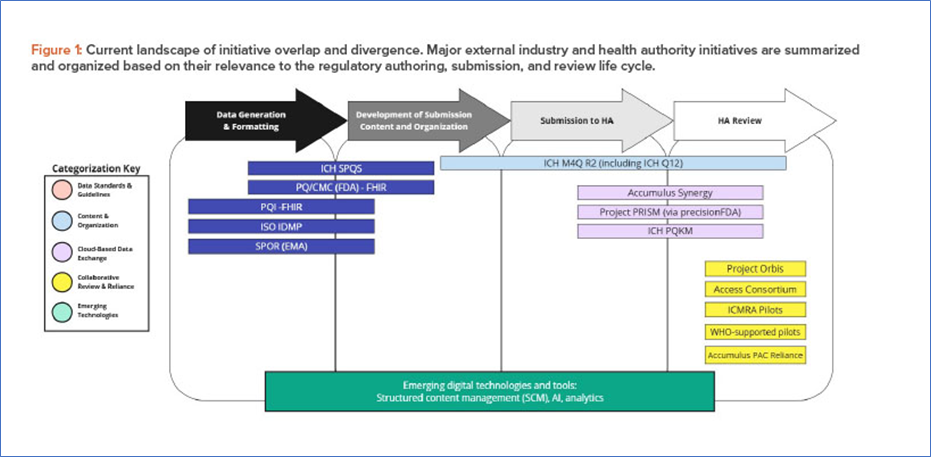
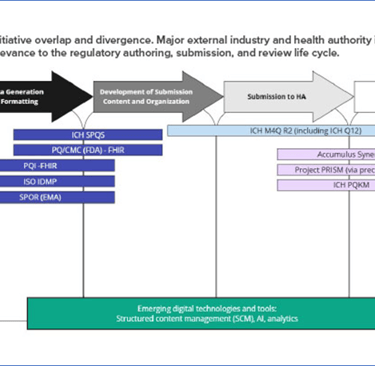
Major initiatives overlap
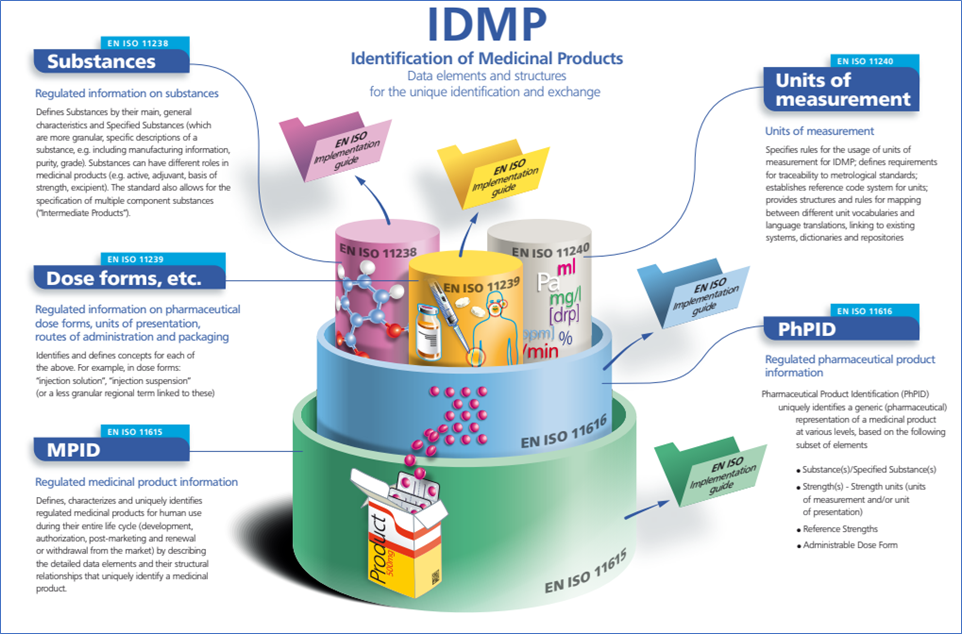
ICH IDMP, Reference: https://isotc215-wg6.team/wp-content/uploads/2018/07/IDMP-flyer_final_light.pdf
IDMP: Overview of ISO IDMP data elements - for illustration purposes only, Reference: https://www.ema.europa.eu/system/files/documents/other/wc500217406_en.pdf
EMA Network Portfolio roadmap, Reference: https://www.ema.europa.eu/en/documents/other/network-portfolio-roadmap_en.pdf
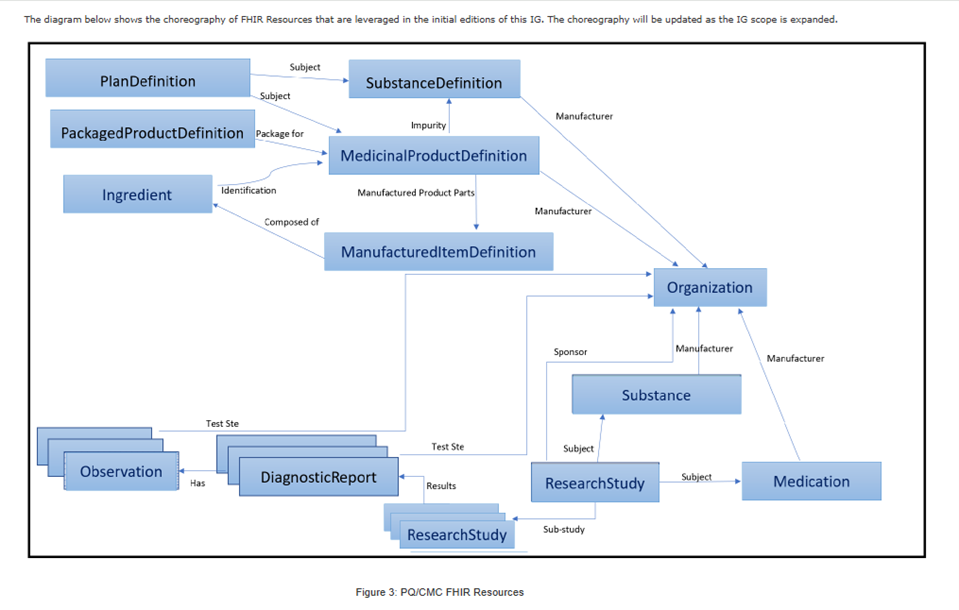
PQ/CMC FHIR resources, Reference: https://hl7.org/fhir/us/pq-cmc-fda/STU2/
PQ/CMC and IDMP High-level Mapping, Reference: https://www.fda.gov/industry/pharmaceutical-quality-chemistry-manufacturing-controls-pqcmc/pqcmc-and-idmp
Digital Data Flow (DDF), Unified Study Definitions Model (USDM), ICH M11 Timeline, Reference: https://www.cdisc.org/sites/default/files/2025-05/7DE%20Digital%20Data%20Flow%20ICH%20M11%20Current%20State%20and%20Aligning%20TMF%20Standards%20Development.pdf
ICH M11 Opportunity, Reference: https://www.cdisc.org/sites/default/files/2024-10/Interchange%20-%20October%20final.pdf
ICH M4Q(R2) to Structured Product Quality Submission (SPQS)(M16), Reference: https://database.ich.org/sites/default/files/M4Q%28R2%29_Step%202_Slides%20to%20Accompany%20Consultation_2025_0624_SEC.pdf

ICH M4Q(R1) vs. ICH M4Q(R2), Reference: https://database.ich.org/sites/default/files/M4Q%28R2%29_Step%202_Slides%20to%20Accompany%20Consultation_2025_0624_SEC.pdf
ISPE Pharma 4.0 ™ Operating Model, Reference: https://ispe.org/initiatives/pharma-4.0
Pistoia Alliance IDMP-ontology, Reference: https://marketing.pistoiaalliance.org/hubfs/2023-07%20IDMP%20Ontology%20Pitch%20Deck%20%281%29.pdf
From Pharmaceutical Engineering, Major initiatives overlap, Reference: https://ispe.org/pharmaceutical-engineering/january-february-2026/cohesive-vision-single-global-cmc-dossier
Contact
(This effort is strictly volunteer, and a work in progress; mobile site has not been optimized for size. The creator welcomes constructive suggestions on how to improve this resource or to correct errors.)
Email: contact@e2digitalpharma.info
© 2026. All rights reserved.

